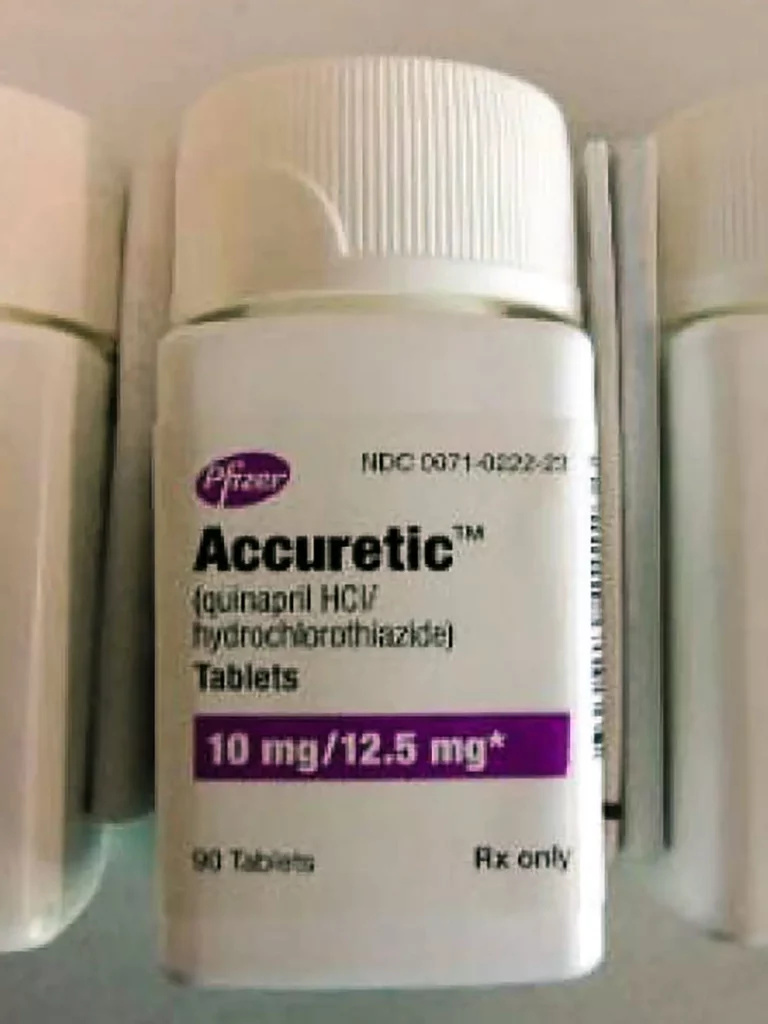
Pfizer is recalling some shipments of Accuretic, a blood pressure medication, as well as authorised generic copies of the drug because a cancer-causing ingredient found in those batches exceeded the permissible daily intake threshold.
The drug contains nitrosamine, which is also found in water, alcohol, and some foods such as bacon and grilled meats.
Patients who use the medications for a long time are thought to have a small risk of cancer.
As private labs and firms have discovered cancer-causing chemicals and triggered widespread recalls, especially in blood pressure medicines, such recalls have become more regular in recent years.
The Food and Drug Administration recommended companies to look for these substances on a regular basis in September 2020.
On Wednesday, the FDA issued another nitrosamine-related recall, this time for a Sandoz medicine used to treat severe musculoskeletal problems.
The Food and Drug Administration (FDA) issued a recall of valsartan-containing drugs in 2018 after NDMA, a possible carcinogen, was identified in them.
Accuretic has a small number of users: approximately 1,300 people took it in 2020.
According to data from IQVIA, an analytics firm, the generic formulation has more than 192,000 consumers.
According to IQVIA data, the generic medicine, which contains the chemicals quinapril and hydrochlorothiazide, generated around $4.7 million in sales in 2020.
In a press release, Pfizer stated that it was unaware of any adverse occurrences linked to the Accuretic recall.
It claimed that the drug’s advantages outweighed the hazards, claiming that it lowers blood pressure and lowers the risk of strokes and heart attacks.
Patients should speak with their doctors about other treatment choices, according to the business.


